The CF Life Sciences team attended the ARUK 2026 annual conference in Manchester this week. It is largest gathering yet, attracting over 700 delegates. Across two days of science, strategy, and clinical data, one message has come through clearly: this field is no longer asking whether disease modification is possible. It is asking how fast, how early, and for whom.
Below we share our key takeaways from both days, from the opening plenary’s strategic overview of the treatment landscape, to the latest clinical data on two of the most promising therapies in development.
Day 1: The Dementia Treatment Landscape
The opening plenary, delivered by Professor Cath Mummery (UCL / NIHR Dementia Translational Research Collaboration), offered one of the most clinically grounded and strategically important overviews of the dementia treatment landscape in recent years. For those working at the intersection of life sciences, healthcare strategy, and health investing, the signals were clear: the field is accelerating, the questions are sharper, and the implications for how we design, fund, and deliver research are profound.
Anti-Amyloid Therapy: From Validation to Optimisation
The approval of lecanemab and donanemab marked a pivotal moment: proof that the amyloid hypothesis is correct and that the disease course can be altered. Yet it was clear that validation is not the destination – it is the starting point. Both agents achieve meaningful amyloid reductions (60–87 centiloids over 18 months) but with modest clinical benefit and a significant safety signal: amyloid-related imaging abnormalities (ARIA), affecting 12–25% of patients on standard monoclonal antibody therapy.
Key message: It is clear that amyloid-targeting therapy works but the next challenge is improving the speed of clearance, reducing side effects, and identifying who benefits most.
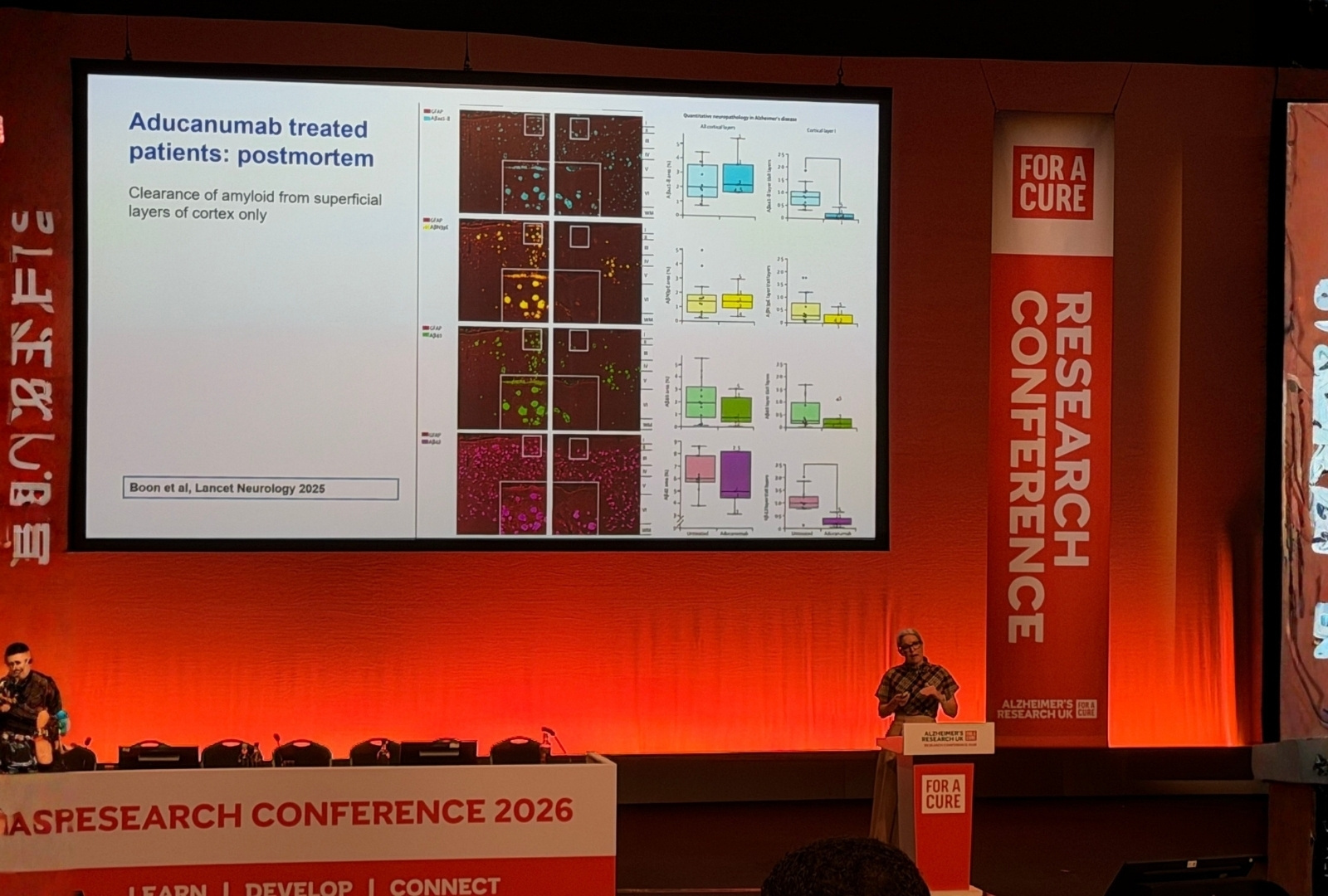
The most striking clinical data concerned trontinemab, a novel compound that actively transports the drug across the blood-brain barrier via a transferrin receptor-binding module. The mechanism matters: active transport via capillaries rather than passive diffusion through large vessels appears to reduce vascular amyloid engagement and therefore ARIA risk.
Key message: Trontinemab achieved a 91 centiloid amyloid reduction in just three months, with 92% of trial participants reaching amyloid-negativity on PET at six months, and an ARIA rate of under 3%.
The Case for Acting Earlier
Post-mortem and biomarker data show that tau accumulation becomes self-sustaining at the 40–60 centiloid amyloid threshold: a ‘forest fire’ that, once ignited, is far harder to contain. The implication is that waiting for clinical symptoms, or even mild cognitive impairment, may already be too late for some patients.
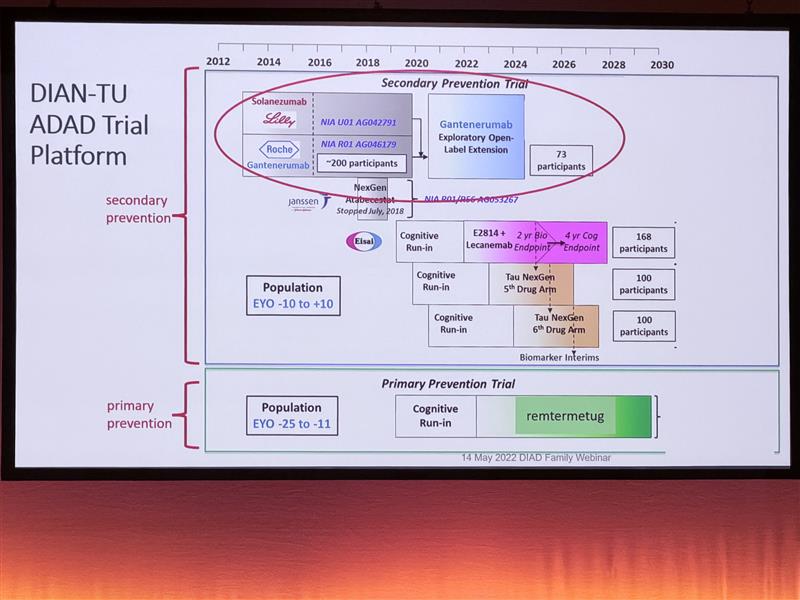
Key message: The DIAN-TU platform trial showed that individuals from genetic Alzheimer’s families who received over ten years of treatment had a 50% reduction in risk of symptom onset, with modelling suggesting treatment may have delayed onset by up to a decade.
Gene Silencing: A New Class of Therapy Enters the Frame
Two candidates stand out. An RNAi therapy (Ionis) targeting APP mRNA reduces amyloid precursor protein production at source, achieving approximately 80% reduction in amyloid precursor fragments from a single dose, sustained over 12 months, and potentially requiring only twice-yearly dosing. The second, ION-464 (Ionis/Biogen), targets tau mRNA and has demonstrated sustained 40% reductions in CSF tau, with a 364-patient Phase 3 study results expected within months.
Key message: Gene silencing therapies targeting both amyloid and tau are now in late-stage trials, and both are being explored in combination with anti-amyloid therapies in the DIAN-TU platform.
The UK as a Global Hub for Early-Phase Trials
Of all European early-phase Alzheimer’s trials currently active, all are based in the UK. The UK Dementia Trials Network is building on this with harmonised site contracting and a growing network of expert sites. Phase 3 site initiation that previously took an average of 175 days is now being achieved in 42 days across UK network sites.
Key message: This is a competitive advantage for pharmaceutical companies considering where to place late-stage investment.
Patient Centricity: The Most Urgent Gap to Close
A survey of over 1,200 people living with dementia found that fewer than 20% were told about research opportunities at clinic, fewer than 3% enrolled in a clinical trial, and only 40% felt they were even able to ask about participation.
Key message: Fewer than 3% of people with dementia in the UK currently enrol in a clinical trial. This is not just an ethical issue, it is a scientific and commercial one that the field must urgently address.
Day 2: Clinical Data Sessions: Trontinemab & Donanemab
Day 2 moved from strategic overview to detailed clinical science, with two presentations providing the latest data on two of the most advanced therapies in the Alzheimer’s pipeline. Both reinforced the same central theme: earlier treatment leads to better outcomes, and the evidence for genuine disease modification is becoming harder to ignore.
Trontinemab (BrainShuttle™ AD): Rapid and Robust Amyloid Removal
Data from Roche provided an update on the BrainShuttle AD Phase Ib/IIa study. The talk providing the most detailed clinical readout yet on trontinemab’s performance in patients with MCI or mild-to-moderate Alzheimer’s disease.
Trontinemab is engineered as a bispecific antibody that binds to transferrin receptor 1 (TfR1) on brain capillary cells, crossing the blood-brain barrier via transcytosis before engaging and clearing amyloid plaques. This results in significantly higher brain concentrations than conventional antibodies, enabling faster and more complete amyloid removal with a lower ARIA risk profile.
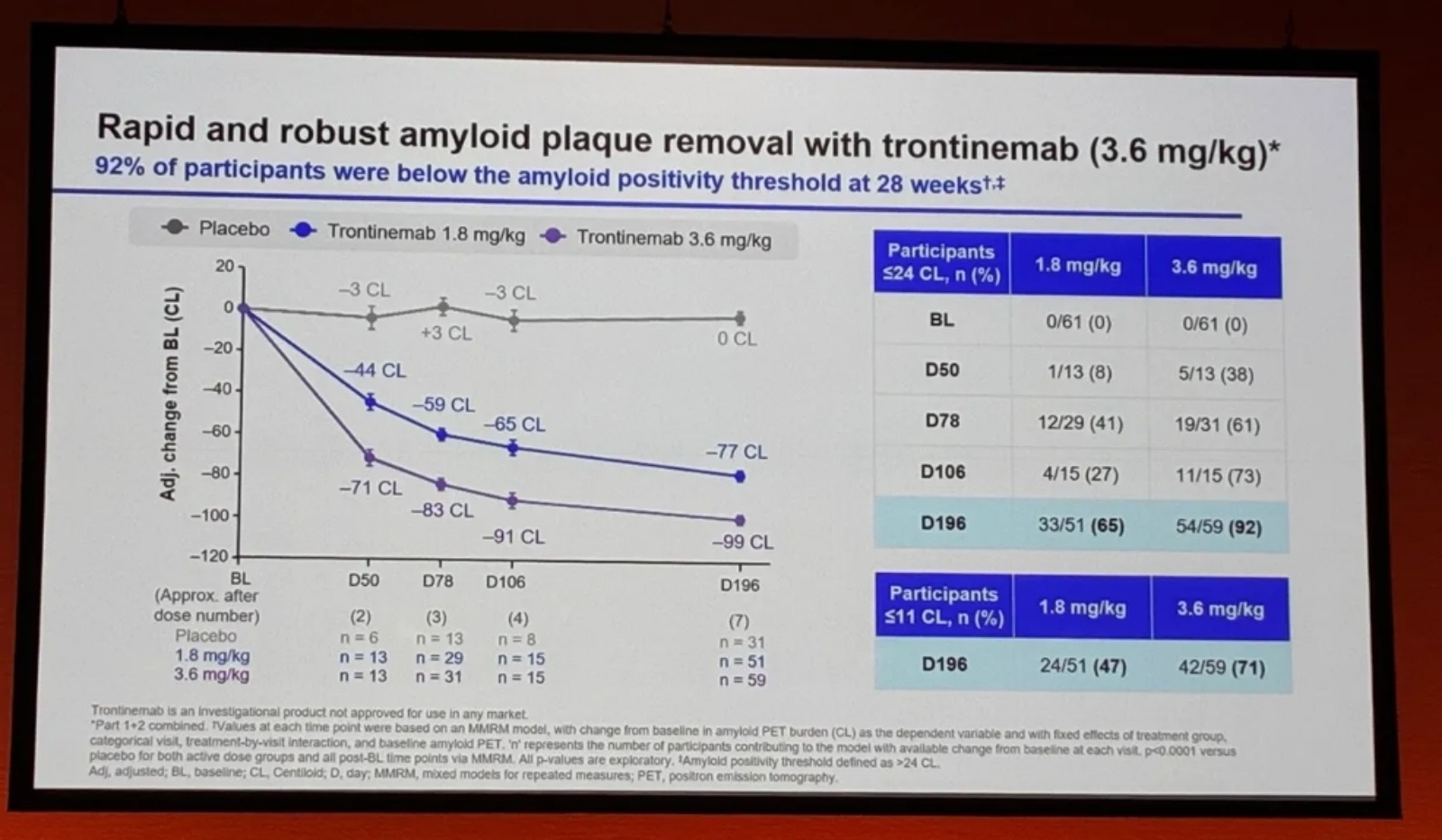
Key message: At the 3.6 mg/kg dose, 92% of participants fell below the amyloid positivity threshold within 28 weeks — and 71% reached levels potentially comparable to those seen in cognitively healthy individuals.
CSF biomarker data showed a 28% reduction in pTau181 and an 8% reduction in MTBR-tau243, a marker linked to tau tangle pathology, suggesting trontinemab’s effects extend beyond amyloid removal to downstream pathological processes. On safety, ARIA-E incidence remained below 5%.
Key message: Two Phase III trials, TRONTIER 1 and TRONTIER 2, are now actively recruiting across 14 UK sites and 18 global regions. A prevention study in preclinical AD (PrevenTRON) has also been announced.
Donanemab (TRAILBLAZER-ALZ 2 LTE): Benefits That Keep Growing Even After Treatment Ends
The second session presented three-year follow-up data from the TRAILBLAZER-ALZ 2 Long-Term Extension (LTE) study of donanemab (Lilly), comparing early start and delayed start cohorts against an external propensity-matched ADNI cohort.
Key message: Donanemab’s clinical benefit continued to grow over three years, even after most participants had completed their active treatment course, consistent with a genuine disease-modifying effect.
Early initiation of donanemab reduced the risk of progressing to the next stage of Alzheimer’s disease by 27% (hazard ratio 0.729, p<0.001) compared to the delayed start group, reinforcing that earlier treatment leads to meaningfully better long-term outcomes.

Key message: Amyloid clearance achieved with donanemab was durable, with plaque reaccumulation estimated at just 2.4 CL/year, comparable to the natural rate, and no new safety signals were identified over three years.
What This Means for Health Investors and Life Sciences Leaders
Across both days, the signals from Manchester are clear and carry real strategic weight. The dementia drug pipeline is diversifying fast, from monoclonal antibodies to gene silencing, from symptomatic management to primary prevention. Combination approaches, earlier intervention windows, and active transport delivery mechanisms are all advancing simultaneously.
Five things that stood out across ARUK 2026:
Thanks to #ARUK 2026 for organising such a thought-provoking conference.
Disclaimer: This blog post summarises independent scientific presentations at the ARUK 2026 conference. Conference sponsors had no influence over scientific content, speaker selection, or the agenda. All abstracts were independently peer reviewed. Presentations reflect the independent views of speakers, not those of sponsoring organisations or AIUK. Trontinemab is an investigational product not approved for use in any market.
CF’s Life Sciences practice work with pharmaceutical companies and healthcare organisations navigating exactly these questions, from market access strategy and health economic modelling to clinical trial site selection. If you would like to discuss what this evolving landscape means for your organisation, we would be pleased to hear from you.
About CF
CF is a leading consultancy dedicated to making an enduring impact on health and healthcare. We work with leaders and frontline teams to improve health, transform healthcare, drive adoption of innovation and create value through investment.
About the authors

Ben Richardson
Ben is Managing Partner at CF and works at the intersection of health systems and life sciences in the application of health data to create insights, enable intervention and generate evidence. He is an expert in population health, the uptake of innovation, real world evidence and the economics of health. Ben leads CF’s Life Sciences and Health Investing practices and Data Sciences capability.
Email [email protected] to get in touch.






